Table of Contents
Introduction
We were extremely disappointed to read this letter authored by the BAD and released on December 6, 2022. Over the last 2 years and as recently as in the last few weeks we have attempted contact with the BAD through:
- Letters to the BAD leadership directly
- Direct requests passed to BAD leadership by dermatologists that we work with
- NHS partner organisations requesting three-way calls
- Open invitation to join our webinar where we shared performance from our post-market surveillance, including a direct invitation sent to the Chair of the BAD AI Working Group
- The NHS Accelerated Access Collaborative who were approached by the BAD
- The NHS National Innovation Accelerator who were approach by the BAD
- Several Academic Health Science Networks we work with
Our only contact with the BAD was in June of this year, we received an email from an academic team working with a small amount of funding from the BAD and requesting our clinical evidence. However once again our request for a call did not materialise though while preparing this letter we received an email on the 13th of December which informed us that the research was outside of the BAD and agreeing to our request for a call.
We see no world where AI is not supporting NHS dermatology pathways in the future. The BAD have constructive concerns that will improve AI based care. However, they are taking a position without attempting to properly inform their position.
Particularly in the last letter, we feel that there are a number of inaccuracies or misleading points and that the following points should be made clearly and on the record. As outlined in our open letter in response, we strongly believe that claims should be backed by evidence so below you will find a detailed document responding to the BAD’s letter with further reasoning and references.
Before we analyse the letter, it is worth providing some context for the state of dermatology nationally and how Skin Analytics is working with our NHS partners.
Cancer Wait Times for dermatology are challenging
Despite the significant and sustained efforts from Dermatology teams across the country, cancer wait time targets are not being met. The Q2 Provisional Provider Cancer Wait Times suggest that only 19% of Trusts were able to see their skin cancer 2WW referrals within 14 days. Nationally, only 62% of patients urgently referred for suspected skin cancer were seen within 14 days while more than 10% of 2WW referrals waited more than 4 weeks 4.
Nationally urgent skin cancer 2WW referrals are increasing more than 11% each year, ignoring the main year of covid impacts 5, and without innovation, we cannot expect our Dermatology teams to be able to achieve these targets in the future.
How Skin Analytics is supporting NHS dermatology
Skin Analytics’ DERM is the only approved Class IIa AI dermatology product in the UK. In addition to being regulated by the MHRA, we have independently audited quality and information management systems. We have post market surveillance data from more than 15,000 patients that shows our AI exceeds the performance targets set by our Clinical Advisory Committee and agreed with our NHS partners.
As many people, including the BAD, are unaware of how our pathways operate, we wanted to share information on the pathways. For simplicity we will focus on secondary care as the majority of our users are post 2WW referral secondary care partners.
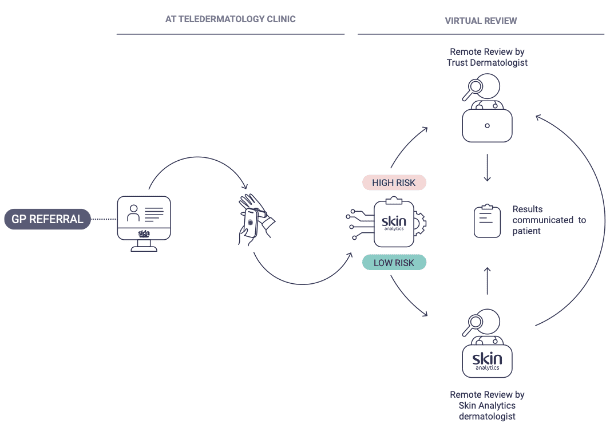
The high level steps are shown below and in the diagram:
- GP refers to secondary care on the 2WW pathway as normal.
- Patients’ first appointments are at an imaging clinic, where consent, medical history and context and dermoscopic images are captured.
- DERM assesses the dermoscopic images of lesions only.
- High risk lesions (Melanoma, Squamous Cell Carcinoma, Basal Cell Carcinoma, Bowen’s Disease and often but not always Actinic Keratosis), or those excluded from DERM assessment are allocated to trust dermatologists to review virtually based on the images and medical history. Dermatologist outcomes are communicated with patients, and any appropriate next steps are arranged.
- OPTION OF SECOND READ – Low risk lesions can be checked by a UK based consultant dermatologist contracted by Skin Analytics:
- If the dermatologist disagrees with DERM’s decision, cases are passed back to trust dermatologists to review and decide on the appropriate treatment for the patient based on local pathways.*
- If the dermatologist agrees with DERM, the case continues to step 6.
- Patients with low risk lesions are sent an outcome letter confirming this and are discharged from the 2WW pathway.
*Note – these cases where a dermatologist overturns a decision to discharge a case by DERM are the source of our sensitivity calculations referenced elsewhere in this document. We track the outcomes of these cases to obtain the final outcome for those cases.
We have significant experience and are achieving good outcomes with our partners
Skin Analytics has now assessed over 38,000 NHS patients for suspicion of skin cancer. During the last quarter, we assessed around 35% more skin cancer patients than the largest skin cancer 2WW receiving Trust in England.
Our last two quarters of performance reporting show that our system consistently performs at a very high level while reducing the number of 2WW referrals dermatology teams need to review face to face.
- In the last reported quarter our first partner, University of Hospitals Birmingham, achieved the highest 2WW performance numbers of any Trust in the UK who saw more than 160 patients in the quarter 6.
- West Suffolk Foundation Trust 2WW performance went from around 20% to 95% (Sep 2021 – Apr 2022).
- University Hospitals Leicester 2WW performance improved from 44% to 91% (May-Aug 2021 vs May-Aug 2022).
A detailed analysis of the BAD’s letter dated December 6, 2022
We believe that the information in the BAD’s letter, “The use of Artificial intelligence (AI) to tackle 2WW referrals” dated 6th December, is either factually incorrect or potentially misleading. Below we will explain why we believe that is the case and justify our position with data where possible.
The letter was about Skin Analytics
Firstly, while did not specifically name Skin Analytics, it is clear that the letter was directed at us. The sites listed in the appendix are all sites where Skin Analytics has worked. Further, the quote provided was from University Hospitals Birmingham where Skin Analytics first deployed an AI solution in early 2020 in response to covid challenges. The letter expresses concern about AI led decision making that is only possible in the UK as a Class IIa UKCA marked device. Skin Analytics is also the only Class IIa UKCA marked AI medical device for skin cancer.
However, we should note that in Cambridge University Hospitals and North West Anglia we only operated an outsourced consultant dermatologist teledermatology service for GPs in Cambridge and Peterborough CCG with no use of AI. Including this as a data point within a letter focussed on the use of AI shows a concerning lack of knowledge about our pathways by the BAD.
BAD Argument 1
The accuracy of AI algorithms intended to support skin cancer diagnoses have been overestimated, with studies conducted in settings that do not reflect the full breadth and depth of clinical practice or the required exposure to over 2000 types of skin disease. This introduces significant bias and risks missing serious but rare diagnoses of skin cancer which can lead to patient harm. Furthermore, the limited classification of skin lesions by an AI technology into either low or high-risk lesions does not significantly reduce the number of referrals from primary to secondary care.
AI performance in skin cancer diagnosis has been overestimated
In some respects we agree with the BAD, but as a broad statement covering all skin cancer AI it is not accurate. Most performance numbers presented by early stage AI companies are from small internal datasets which are often misleading. Further, the literature suggests that many AI systems’ performance decreases when moving from observational to prospective data and this holds across a range of health specialities.
However, Skin Analytics has conducted prospective clinical studies and has significant amounts of real world data which shows what actually happened to patients at NHS sites using our technology. As our longest running partnerships we have the most data from University Hospitals Birmingham Foundation Trust (UHB) and West Suffolk NHS Foundation Trust (WSFT).
We report quarterly on performance. The data below covers patients assessed by the most recent version of DERM up until August 2022. These results are from the “messy reality in clinics” our BAD colleagues refer to 7.
UHB: ~ 2000 lesions assessed by DERM during period assessed
- 100% (161/161) sensitivity for skin cancer, including 47 melanoma, 44 SCC, 69 BCC and 1 rare cancer
- 99.8% (1,155/1,157) skin cancer negative predictive value (NPV) (2 BCCs were referred as pre-malignant lesions based on the pathway agreed locally)
WSFT: ~ 500 lesions assessed by DERM during period assessed
- 100% (62/62) Sensitivity for skin cancer – including 17 melanoma, 13 SCC, 31 BCC and 1 rare cancer
- 100% (270/270) skin cancer negative predictive value (NPV)
In summary, since April 2020, we have been running AI dermatology services and collecting outcome data from the messy reality of 2WW skin cancer clinics. We are performing well above the 95% sensitivity target that was set by our Clinical Advisory Committee and agreed with our NHS partners. By any statistical method, we have a very precise understanding of how our system performs with our NHS partners.
The clinical evidence for AI is not sufficient
AI used in skin cancer pathways are medical devices and therefore governed by law under The UK Medical Device Regulations 2002 (“UK MDR”) which is overseen by the MHRA, the UK’s medicines and healthcare products regulatory agency. Medical device regulation requires that manufacturers like Skin Analytics identify the benefits and risks to patients and provide sufficient evidence that residual risks are sufficiently low and outweighed by the benefits of the medical device.
The classification of medical devices under the UK MDR is a risk-based system taking into account the intended use and potential risks associated with a medical device. This approach uses a set of ‘classification rules’ to determine the appropriate classification for a device and, in turn, indicate the level of regulatory control and oversight required for the device.
Our AI product is classified as a Class IIa medical device. This means that our device is required to undergo a comprehensive quality and technical assessment by an independent body appointed by the MHRA to determine whether the device is compliant with the UK MDR. A key component of this assessment is the independent review of our Clinical Evaluation Report (“CER”). The CER is a report that identifies, appraises and evaluates clinical evidence for a device in order to verify the safety and performance, including clinical benefits, of the device when used as intended. Skin Analytics has successfully completed this rigorous assessment and our device remains the only Class IIa approved AI medical device in dermatology.
Securing a Class IIa medical device is not a milestone, it is a commitment to continue to operate an effective quality management system that is evaluated against the best in class standards agreed by regulatory bodies. That means that we are constantly working to maintain our medical device and operate safe and effective processes. It is a living process and one in which we are constantly looking for improvement. AI as a medical device should be held to high standards and that is what the regulatory requirements do both with respect to clinical evidence and as importantly the quality and safety systems a company operates.
Specifically with respect to our clinical evidence, we have followed the guidelines of ISO14971 which outlines the required approach to risk management. The most obvious risk is the ability, or inability to find malignancies in skin lesions, this is the sensitivity of our test. We have conducted prospective studies to evaluate this and are continuously collecting outcome data on patients that we assess to build on that evidence.
While there are many other risks we identify and manage, the other obvious risk is whether in finding sufficient malignancies, the system causes patient harm through over referral. While clearly sending healthy patients to our dermatologists carries a lower risk to patients, this is critical all the same. Over referrals may overwhelm dermatology teams, slowing the system down and causing cancers to be untreated for longer. This is what we are seeing right now at alarming levels but it is not as a result of the use of AI.
We have assessed this risk in clinical studies and validated with real world evidence that this isn’t the case. From our live deployments we see that we are able to discharge more than 70% of benign lesions referred under the 2WW with the latest version of our medical device 8.
We know this because as mentioned earlier, we monitor performance very closely with a detailed multi level Post Market Surveillance Plan that includes our Real World Evidence Protocol. This allows us to produce the data summarised above and in provided in more detail in our quarterly performance reports which are shared with partners. It’s worth noting that we presented this data on a webinar in September to which we invited members of the BAD AI working group. These report include:
- AI performance versus gold-standard diagnosis at the end of the patient’s journey through the system (wherever this data is made available by our partners).
- Number of patients who present multiple times for the same lesions and any instances where a worse outcome is detected on the second presentation.
To the best of our knowledge, this far exceeds the level of active monitoring which takes place in traditional services and taken together there is sufficient evidence to support the pathways we have built with our partners.
An AI not able to identify all of the more than 2,000 skin conditions risks introducing significant bias and missing serious but rare skin cancers
DERM is being used within patient pathways to evaluate skin lesions that are either about to be, or have been referred on a 2WW pathway for suspected skin cancer. In the 2WW pathways, national data suggests that around 8% are malignant lesions that need urgent attention 9.
The evidence shows DERM is very good at identifying malignant lesions and therefore enabling the discharge of benign lesions from the urgent referral pathway. It has not been designed, nor does it need to identify all skin conditions to successfully help our partners deal with urgent referral demand.
We didn’t build DERM to replace dermatologists. The reality is that there is a global shortage of specialists in dermatology. If this wasn’t the case, there would be no need for DERM. Medicine is an intrinsically human science and will always be so. To imply that any AI system should be able to replicate all of what we train our specialists to do is just not accurate and suggests a concerning misunderstanding of the role AI can play to support the known pressures on Dermatology departments.
We have built a system specifically designed to address the challenge of skin cancer. This means assessing a large number of healthy patients to find those with cancer. AI is very good at this both objectively and in comparison to clinicians where the literature suggests that specialists finds less than 92% of the melanoma they see on average 10.
We are constantly amazed by the breadth and depth of the knowledge of our clinical colleagues and this knowledge goes far beyond picking malignant lesions from benign ones. However, given almost half of specialists’ time is spent on skin cancer 11, we’re confident AI has value even if it isn’t trying to identify each of those more than 2,000 skin conditions.
The use of AI does not reduce the number of referrals from primary to secondary care
This point is made again by the BAD as the final point of the position statement. We will address it in this later section. However, when deployed post 2WW referral, AI is not in a position to reduce the number of referrals from primary to secondary care. Rather AI can support dermatology teams to identify cancers by discharging benign lesions.
BAD Argument 1. Summary
Skin Analytics works with the MHRA, our UK Approved Body and each of our partners to evaluate our evidence, the quality management systems we have in place, our service’s clinical safety and our processes for delivering services to patients. We have met every requirement to operate our services and as shared within this response, we have delivered positive results with our partners.
Despite having very strong evidence to support the use of DERM within the pathways we have deployed, we want to stress that no AI system is perfect. Bias and patient risk needs to be a detailed and continual conversation and one held within close partnerships so that both can be reduced as far as possible. Unfortunately it is not a discussion the BAD have been willing to participate in.
One existing aspect of patient risk that can’t be ignored is the large backlog of patients waiting to see dermatologists. There are serious skin cancers waiting to be discovered and these aren’t just in the 2WW patients that are being prioritised. Without support to reduce waiting times, the impact on patients may well be very significant.
We call on the BAD to join a conversation to quantify the relative risk of using an AI system with performance numbers validated with a large number of British patients against the risk of the impact of the current wait times. Without engaging in this discussion, any position against AI is uninformed.
BAD Argument 2
Using AI in primary care does not reduce the need for a Consultant Dermatologist to confirm that the low risk (benign) classification is correct. Onward referral of high-risk lesions will include a significant number of pre-cancerous and non-malignant skin cancers. Using nationally recognised teledermatology 2WW models would have triaged these patients the first time to the correct pathway or discharged care back to their GP. These models are in the best interest of clinicians and patients.
Using AI in primary care doesn’t reduce the need for a Consultant Dermatologist to confirm the lesion is benign
There is no requirement for a consultant dermatologist to review an AI’s output. Appropriately regulated AI can support consultants, GPs, or even work autonomously with no healthcare professional’s input, providing patients give appropriate informed consent.
We assume from this statement that the BAD is conflating our use of a consultant dermatologist as a second read of the AI as a requirement. In our deployments we generally have used consultant dermatologists to review low risk lesions and originally implemented this model to re-assure partners of the safety of using DERM. We intend to remove the measure over the coming year in consultation with our partners.
We expect this misunderstanding stems from the BAD refusing to meet with Skin Analytics to understand our pathways, evidence and risk controls. Again we extend another open invitation to the BAD to understand the use of AI within the NHS.
Onward referral of high-risk lesions will include a significant number of pre-cancerous and non-malignant skin cancers. Using nationally recognised teledermatology 2WW models would have triaged these patients and is in the best interests of clinicians and patients
The BAD are correct that despite DERM being able to safely discharge more than 70% of benign lesions from 2WW referrals 12, there are still a number of lesions which turn out to be non-melanoma skin cancers, premalignant lesions or benign lesions that DERM sends to the dermatology teams.
This is an important risk control, recognising that we place a significant priority on identifying skin cancers over discharging benign lesions as discussed earlier. It is worth noting that clinicians themselves use the same risk control and on average globally dermatologists biopsy more than 7 lesions for every melanoma they find 13.
With this said, it is not clear what evidence the BAD are using to conclude that using teledermatology triage model would significantly reduce the number of lesions being assessed by dermatologists.
Teledermatology is recognised as being an accurate way to diagnose skin cancers with less than 7% of cancers being missed according to a recent Cochrane review 14. Ultimately, it is clear that teledermatology is a powerful tool in dermatology and the NHS has done an excellent job to create an environment to adopt the innovation safely.
However, the efficiency gains of teledermatology alone do not appear to be sufficient to resolve capacity issues. Nor is it clear why the two approaches are not complementary. Accruing the benefits of both the discharge of benign lesions from within 2WW referrals by AI and teledermatology reviews of the remaining lesions by consultant dermatologists would be positive outcomes for both patients and clinicians and is what we deliver with our NHS partners.
In every instance that Skin Analytics has deployed DERM, we do so within our teledermatology platform which enables clinicians to remotely review patients and decide appropriate next steps for those patients.
BAD Argument 3
In secondary care it is a legal requirement for patients referred on a 2WW pathway to be seen (virtually or face to face within Cancer Waiting Times (CWT) guidelines) by a consultant (or member of their team) who are all core members of the local hospital skin cancer multidisciplinary team (LSMDT) and/or specialist skin cancer multidisciplinary team (SSMDT). There is no legal remit for AI to be used independently (i.e., outside of this pathway) to triage or diagnose a 2WW referral. If AI is used within this pathway, it increases costs as duplicate images are required to be taken by the medical photographer for the patient record and separately for the AI App commercial company. We are aware that the University Hospital Birmingham (UHB) AI model is being used as an exemplar for use of AI, below is a quote provided from the consultants at UHB to put this into perspective.
In secondary care it is a legal requirement for patients referred on a 2WW pathway to be seen by a consultant and there is no legal remit for AI to be used independently
We are not aware of any legal reason why DERM cannot be used autonomously in a skin cancer 2WW pathway and we have previously taken legal advice on this point. The BAD weren’t clear what “legal requirement” they referred to and we would encourage them to clarify this point. We believe they intended to refer to Patients’ rights to be seen within two weeks for cancer referrals under the Handbook to the NHS Constitution.
To first address the intent of the requirement, we are passionate about ensuring that patients can be seen quickly and safely for suspected skin cancer. As we set out above, performance against skin cancer 2WW targets nationally is poor and declining. We have built our AI and service to ensure that patients are safely seen as fast as possible to improve cancer outcomes. We have shared some data on our performance earlier and continue to monitor that we are achieving the intent of patients rights.
To address the detail of the requirement, there is detailed NHS guidance regarding when the two week timeline can end that includes “the patient is seen at a diagnostic clinic or goes ‘straight to test’ in a consultant-led service (unless that test is a blood test)”. NHS Transformation have confirmed that capturing dermoscopic and macroscopic images by a member of the specialist dermatology team meets this requirement (not the assessment of those images).
Our AI, DERM, meets all the regulatory requirements for operating within a pathway comprising autonomous patient assessment. It is classified and independently assessed by a UK Approved Body as a class IIa device under rule 10 as required by Annex IX of Directive 93/42/EEC, which is transposed into UK law through The UK Medical Device Regulations 2002. Rule 10 is for ‘active devices intended for diagnosis’. The usage of DERM within patient pathways are all agreed with local consultants to ensure they are happy with patient care.
If AI is used within this pathway, it increases costs as duplicate images are required
There is no requirement for duplicate images because of the use of AI. Our technology runs on images that are also suitable for review by human clinicians and is captured on hardware approved by the BAD 15 16 . In our case, we have made the decision to restrict the types of image capture devices (cameras and dermatoscopes) that we use with our AI to ensure that we have appropriately tested performance using these devices. The same images that are used for our AI assessment can be used by clinicians.
In some cases, our partner dermatologists request their teams to capture additional DSLR images and of course we support them to do this.
The University Hospital Birmingham (UHB) AI model is being used as an exemplar
While we would be delighted to acknowledge that the BAD recognise our partnership with UHB as an exemplar for AI adoption, the full statement from the BAD is:
We are aware that the University Hospital Birmingham (UHB) AI model is being used as an exemplar for use of AI, below is a quote provided from the consultants at UHB to put this into perspective.
“University Hospitals Birmingham (UHB) have been using AI in the 2WW pathway since April 2020. The clinicians recognise the potential of AI and are keen to continue to work towards developing this in the future. UHB are currently auditing their pathway in order to assess whether AI leads to a reduction in 2WW referrals, cost or clinical time. At present the AI software is being further evaluated and developed and is not yet being used as an independent diagnostic tool.”
We are very proud of our pioneering work and partnership with UHB. Together we designed and deployed post 2WW referral hubs at covid safe sites within 6 weeks to launch them in April 2020. Since Q1 2021 UHB has achieved their skin cancer 2WW targets in every quarter. Over that time, our quarterly performance report shows that we found 98.8% of skin cancers (913 of 924) and since April 2022 we’ve found 100% of skin cancers (223 of 223).
As we mentioned earlier, the latest provider reported provisional data, UHB had the highest percentage of patients seen within 14 days of any provider who had seen more than 200 patients in the quarter and despite receiving the second most 2WW referrals for skin cancer in the country.
We continue to work with a multidisciplinary team within the trust to refine and improve the use of DERM, including exploring the use of DERM in primary care as part of the NHS AI in Health and Care Award. We hope that providing more information around our partnership with UHB gives more context to evaluate the quote the BAD sourced for this letter.
BAD Argument 4 – Health Economics
The BAD has commissioned a report from the University of York on health economic [sic] and the cost-effectiveness of the AI tool across the entire patient pathway. The outcomes of these examples show the potential for additional costs to be incurred for both primary and secondary services due to an increase in 2WW referrals.
It is our mission to help improve skin cancer survival in a way that is sustainable to the NHS. We welcome health economic evaluations in healthcare to ensure that we achieve that goal and to support this we appointed a globally respected health economic researcher to our Clinical Advisory Committee in 2019.
In 2020 we worked with Imperial College and also with the York Health Economics Consortium (YHEC) on health economic evaluations. The resulting health economic models (based on data before the launch of our AI service) suggested that the NHS stood to save money with the adoption of AI technology.
Today, we are undertaking further health economics work based on real world data from live clinical pathways. We are working with the University of Exeter as well as an independent review of the impact of our services as part of the AI in Health and Care Awards by the Technology Specific Evaluation Team (Unity Insights & the University of Surrey). When we conclude these current workstreams, we will be able to share the results, assumptions and models for scrutiny.
Turning our attention to the paper commissioned by the BAD, it is not possible to draw any conclusions from the paper that should impact decision making. The paper set out clearly the potential for AI to impact cancer detection, referral rates and downstream morbidity costs. However, it lacked any details of the modelling undertaken, or cost impact from the above impacts. Furthermore, it was based on assumptions that are not accurate. Specifically, the reported cancer prevalence data in the general population does not match national data, and we reject the premise that an AI should have lower specificity than GPs. Our experience is that DERM can safely discharge a significant percentage of patients on 2WW pathways which have already been deemed concerning by GPs.
BAD Argument 5 – 2WW referral increases
We have also reviewed the 2WW published data for hospitals that have been using AI skin cancer apps or have had this commissioned in their primary care skin cancer pathway. These figures show above average yearly incremental increases in 2WW referrals without significant reductions in inappropriate lesion case mix. The tables are provided in the accompanying appendix.
We’re not clear what point the BAD are making here. We are also unsure how implementing AI in a secondary care setting could logically be expected to reduce referrals made by GPs in primary care who do not have access to this AI. Regardless of this point, rising referral volume without a decline in inappropriate lesions being referred indicates that more cancer is being found which should lead to better patient outcomes.
More importantly, no data presented supports the statement that the AI is increasing referral rates into secondary care. The rising skin cancer referral rate is a long term trend that exists nationally, see data below from Cancer Research UK.
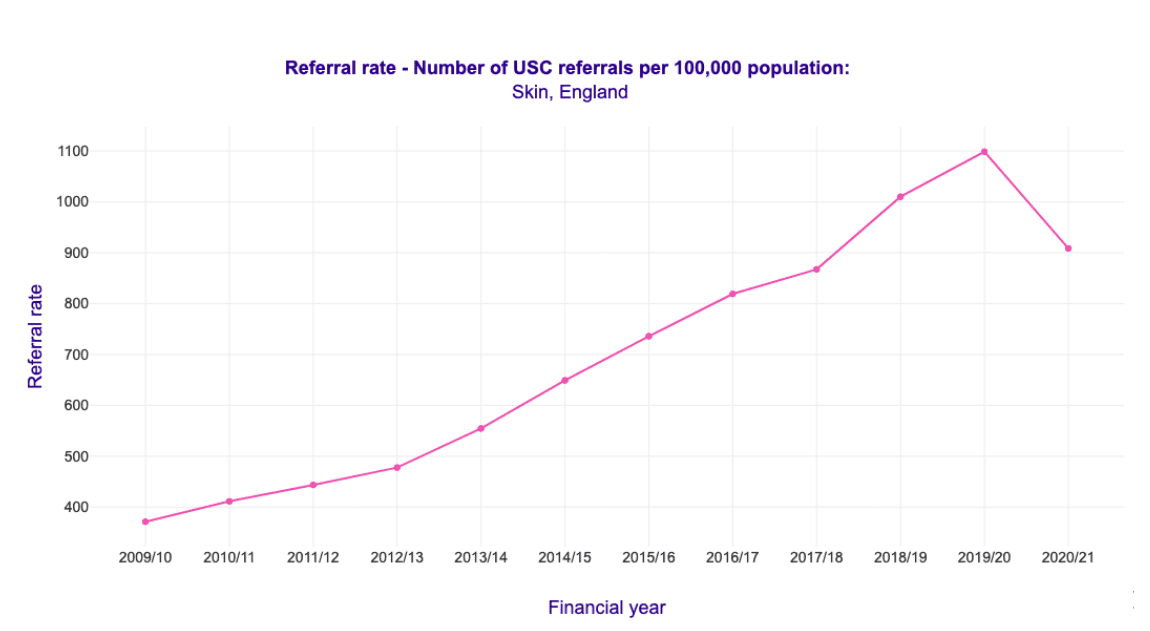
Given the BAD inferred that the use of DERM was the driver for the increase in 2WW at the sites where we are working, below is the extrapolation of that Cancer Research data to those tables which clearly shows this inference is incorrect and misleading.
For this table, we applied the annual growth rate, excluding 2020/21 due to covid, and ignored the effect of population growth to determine expected 2WW volume for 2021/22 based on the long term trends. As is clear below, every Trust with the exception of Chelsea and Westminster Hospital is below the projected referral volume. Incidentally, the data presented for Chelsea and Westminster Hospital is from before AI was used at the site.
2WW Referral Numbers | |||
|---|---|---|---|
| 2019-2020 | ACTUAL 2021-2022 | PROJECTED 2021-2022* | |
| MID AND SOUTH ESSEX NHS FOUNDATION TRUST | 13,876 | 16,359 | 17,236 |
| CAMBRIDGE UNIVERSITY HOSPITALS NHS | 5,518 | 6,507 | 6,854 |
| NORTH WEST ANGLIA NHS FOUNDATION | 4,057 | 4,985 | 5,039 |
| CHELSEA AND WESTMINSTER HOSPITAL | 4,120 | 6,276 | 5,117 |
| UNIVERSITY HOSPITALS BIRMINGHAM | 9,781 | 11,369 | 12,149 |
As a final factual correction, Cambridge University Hospitals and North West Anglia, operated a dermatologist based teledermatology service for GPs without using AI at all.
There is currently no published and independently verified evidence to support the assertion that deploying AI into dermatology pathways increases 2WW referrals within the NHS. This should immediately raise a red flag about both the BAD’s analysis and their understanding of the pathways we have deployed with our partners.
Our Conclusion
The BAD believes that in the future AI will have the potential to improve clinical care in dermatology within an appropriately regulated and governed use. As you are aware, patient pathways in dermatology are complex and to evaluate AI interventions requires clinically led robustly designed research studies. As current technologies cannot address the unmet need of increasing 2WW referrals, we recommend significant local investment in teledermatology services to provide immediate and sustainable results to meet the needs of local patients. More information about this can be found in the NHS demand and capacity management section of our website.
We applaud the BAD’s acknowledgement in the conclusion to their letter that AI will improve clinical care. We disagree that this is resigned to an undefined future date and have laid out our refutations to the BAD arguments against AI with supporting data.
We acknowledge that dermatology patient pathways are complex and robust evidence is required to support the use of AI. However, we reject the assertion that AI is still in the research phase. After nearly 11 years, we believe that we have collected sufficient clinical and real world evidence to support the pathways we deploy with our partners.
In implementing these pathways, we invested in robust risk controls to ensure patients received the right care and as importantly we have been collecting detailed outcome data which we share openly. We believe that the BAD have not appropriately considered the risks that patients face by not adopting AI technology in the face of significant pressure on dermatology teams nationally to achieve Cancer Wait Times.
Patients and medical professionals deserve an open and complete dialogue about how to use AI in dermatology. While the BAD continues to publish letters that are not factually correct or may be misleading, patients and their member dermatologists will continue to miss an opportunity to leverage the UK’s leading position with AI in healthcare.
The Skin Analytics Team
References
4 Q2 2022-2023 Cancer Waiting Times Provider Workbook Provisional
5 Cancer Research UK Early Diagnosis – USCR – Referral rate
6 Provider – based Cancer Waiting Times for Q2 2022/23 Provisional
7 NOTE: We do not claim the system is 100% sensitive for melanoma or cancer. However in the reporting period these numbers are accurate
8 Skin Analytics data contained in Quarterly Performance Reports
9 NHS Cancer Data: Conversion and Detection
10 Visual inspection for diagnosing cutaneous melanoma in adults – Dinnes, J – 2018 | Cochrane Library
11 The crisis in dermatology | The BMJ
12 Skin Analytics data from Quarterly Performance Reporting
15 UK Guidance on the use of mobile photographic devices in dermatology.pdf
16 Dermatoscope Comparison table.pdf
17 Skin Analytics Quarterly Report data – DERM v3.0 and DERM v3.01 combined 15 Jul 2021 – 19 Aug 2022. Available on request
18 Skin Analytics Quarterly Report data – DERM v3.01 15 Jul 2021 – 21 Apr 2022. Available on request
19 Provider – based Cancer Waiting Times for Q2 2022/23 Provisional